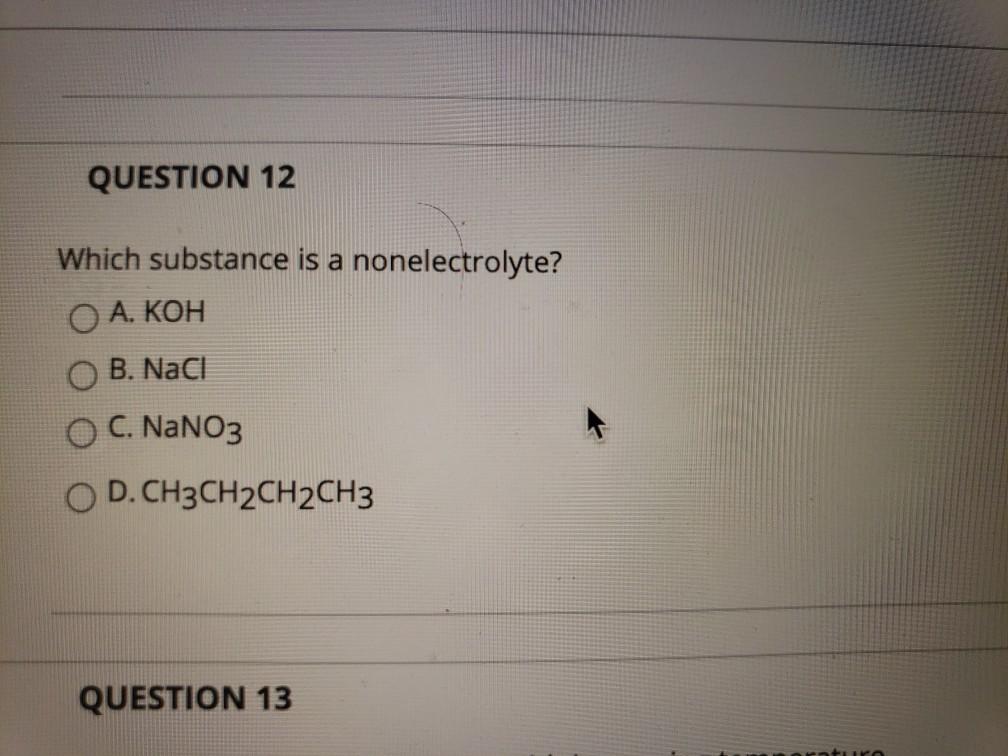
Which Compound Is A Nonelectrolyte . Which substance is an electrolyte ccl4? Therefore, we say that glucose is an example of a nonelectrolyte.
Solved Which One Of The Following Compounds Is A | Chegg.com from www.chegg.com
When these compounds dissolve in water , they do not produce ions. While ccl4, c2h6, and h2o are covalent.
Solved Which One Of The Following Compounds Is A | Chegg.com
A strong electrolyte completely ionizes when dissolved in water. Sodium chloride is an ionic compound that is made up of the sodium cation and the chloride anion. Many molecular compounds, such as sugar or ethanol , are nonelectrolytes.
Source: www.chegg.com
For example, carbon tetrachloride ccl4 when dissolved in water does not dissociate into ions and therefore, it does not conduct electricity. All the others are electrolytes; The answer is (3) hcl.
Source: www.clutchprep.com
While ccl4, c2h6, and h2o are covalent. Acetic acid (ch 3 cooh), the compound in vinegar, is a weak electrolyte. Hcl, hbr, hi, hno 3, h 2 so 4, hclo 4, hclo 3
Source: www.chegg.com
They retain their molecular structure. 1) kcl (aq) 2) h2so 4 (dil) 3) ccl 4 (l) 4) ch 3 cooh (aq) class 10 icse. While ccl4, c2h6, and h2o are covalent.
Source: www.youtube.com
Include soluble ionic salts, strong acids, and bases a. A nonelectrolyte is a compound that does not conduct an electric current in either aqueous solution or in the molten state. Active 2 years, 3 months ago.
Source: www.chegg.com
The definition of electrolyte is a substance which forms ion in an aqueous solution. It does not provide ions in a solution and therefore current does not flow through such solution. A nonelectrolyte is a compound that does not conduct an electric current in either aqueous solution or in the molten state.
Source: brainly.com
A common example of a nonelectrolyte is glucose, or c6h12o6. One of these compounds is an electrolyte, meaning that it. A strong electrolyte completely ionizes when dissolved in water.
Source: www.numerade.com
A nonelectrolyte is a substance that does not exist in an ionic form in aqueous solution. Classify each compound as a strong electrolyte, weak electrolyte, or nonelectrolyte. Strong electrolyte weak electrolyte nonelectrolyte.
Source: wps.prenhall.com
Active 2 years, 3 months ago. When these compounds dissolve in water, they do not produce ions. Conduction of electricity in liquids
Source: www.chegg.com
Conduction of electricity in liquids They share electron pairs to form such bonds. A substance whose aqueous solution conducts no better than water itself is called a nonelectrolyte.
Source: www.focuskimia.com
When these compounds dissolve in water, they do not produce ions. Many molecular compounds, such as sugar or ethanol, are nonelectrolytes. A common example of a nonelectrolyte is glucose, or c6h12o6.
Source: www.theengineeringknowledge.com
Some examples are oxygen, o2, ethanol, c2h5oh, and sugar, c12h22o11. Nonelectrolytes are compounds that do not ionize at all in solution. For example, carbon tetrachloride ccl4 when dissolved in water does not dissociate into ions and therefore, it does not conduct electricity.
Source: www.kentchemistry.com
Many molecular compounds, such as sugar or ethanol, are nonelectrolytes. Salt, on the other hand, is a brilliant electrolyte. Classify each compound as a strong electrolyte, weak electrolyte, or nonelectrolyte.
Source: www.clutchprep.com
Nonelectrolytes do not dissociate into ions in solution; One of these compounds is an electrolyte, meaning that it. When these compounds dissolve in water , they do not produce ions.
Source: www.chegg.com
The below represents a solution of ethylene glycol on the left and sodium sulfate on the right. Sodium chloride is an ionic compound that is made up of the sodium cation and the chloride anion. A nonelectrolyte is a compound that does not conduct an electric current in either aqueous solution or in the molten state.
Source: www.chegg.com
Many molecular compounds, such as sugar or ethanol, are nonelectrolytes. Classify each compound as a strong electrolyte, weak electrolyte, or nonelectrolyte. Therefore, we say that glucose is an example of a nonelectrolyte.
Source: slideplayer.com
This phenomenon is also the reason why solutions containing sugar do not conduct electricity. Those that follow the rules of being soluble in solubility rules b. Is acetic acid a nonelectrolyte?
Source: www.chegg.com
Sodium chloride is an ionic compound that is made up of the sodium cation and the chloride anion. They share electron pairs to form such bonds. 2.the density of the solution.
Source: www.numerade.com
The below represents a solution of ethylene glycol on the left and sodium sulfate on the right. 1.the density of the solute. Conduction of electricity in liquids
Source: www.numerade.com
We’re being asked to classify each given compound as a strong electrolyte, weak electrolyte, or nonelectrolytes.recall that: All the others are electrolytes; Salt, on the other hand, is a brilliant electrolyte.
Source: studylib.net
Which substance is an electrolyte ccl4? Now up your study game with learn mode. One of these compounds is an electrolyte, meaning that it.