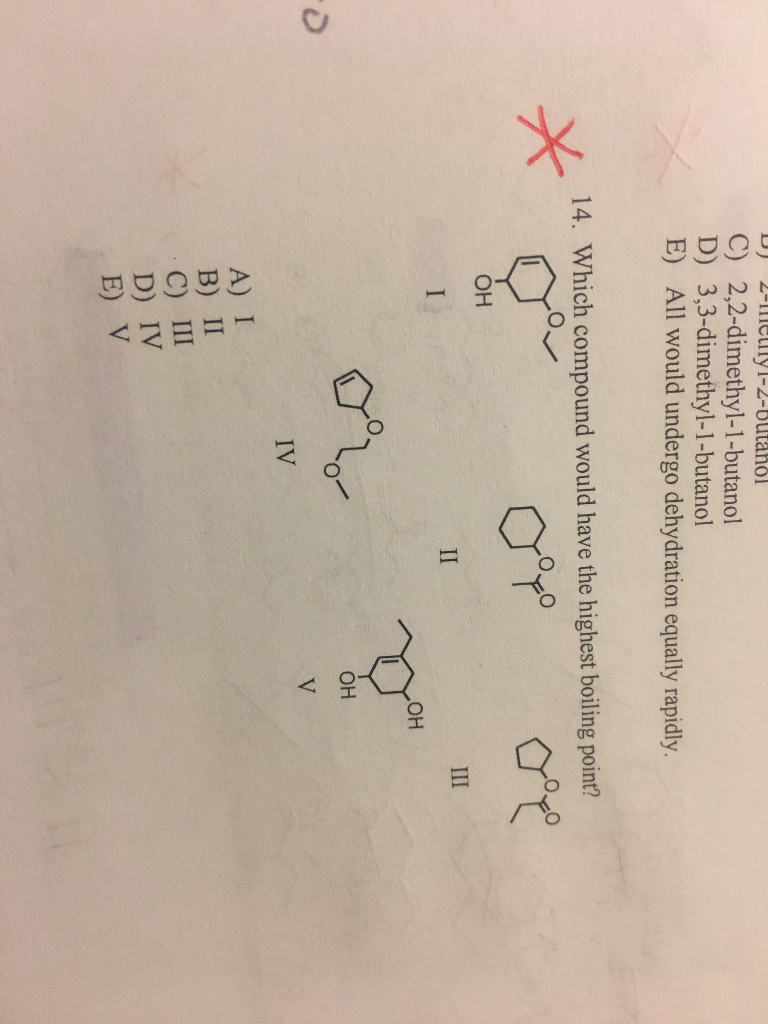
Which Compound Would Have The Highest Boiling Point . That's why they have high boiling point than those of ester. The reason that longer chain molecules have higher boiling points is that longer chain molecules become wrapped around and enmeshed in each other much like the strands of spaghetti.
Multiple Choice Questions Part 2: Funktionella Grp from studylib.net
Due to ability of making hydrogen bonds, boiling point of ethanol is high. For this problem we are asked to determine which of the following compounds would have the highest boiling point.
Multiple Choice Questions Part 2: Funktionella Grp
Therefore the element with the greatest total number of electrons will have the highest boiling point (iodine) and the element with the smallest total number of electrons will have the lowest boiling point (hydrogen). B) ch 3 ch 3. If you look at the molecular structure, we have a chain of 3 carbons, then both molecules differ in number of hydrogen and oxygen atom in the compound.
Source: studylib.net
Water has a simple molecular structure. That's why they have high boiling point than those of ester. However, each of the hexanes has a unique boiling point.constitutional (structural) isomers:
Source: www.toppr.com
# carbons acyclic alkane # of isomers 4 butane 2 5 pentane 3 6 hexane 5 seven heptane 9 6 contents hide 1 which isomer. Further, the boiling point of these compounds follows the order : Of the diatomic elements (h2, n2, o2, f2, cl2, br2, i2), all have dispersion forces.
Source: www.numerade.com
Due to strong intermolecular forces, the molecules of hf will be tightly packed in a lattice. Which compound has the highest boiling point quizlet? The vander waals dispersion forces increase as the length of the hydrocarbon chain increases.
Source: www.chegg.com
If two compounds with same molecular weight then there are different factors which determine the boiling point of the organic compounds. The vander waals dispersion forces increase as the length of the hydrocarbon chain increases. 80% (217 ratings) sign up for free to view this solution.
Source: www.coursehero.com
How can you tell that water has a simple molecular structure? The vander waals dispersion forces increase as the length of the hydrocarbon chain increases. Now both compounds do have hydrogen bonding because h attachment to a high electronegative element, which means boiling point is.
Source: www.clutchprep.com
Due to higher molecular mass. Hydrogen bonding is the strongest intermolecular force. More energy is needed to separate them than short chain molecules which have only weak forces of attraction for each other.
Source: oneclass.com
Increases by increasing the chain length by one carbon. If you googgled, you may find boiling point of $\ce{hf}$ is $\pu{19.5 ^{\mathrm{o}}c}$, while that of $\ce{hi}$ and $\ce{hbr}$ are $\pu{−35.4 ^{\mathrm{o}}c}$ and $\pu{−66 ^{\mathrm{o}}c}$, respectively. The simplest reason is $\ce{f}$ the highest electrnegative atom and capable of having strongest hydrogen bonding.
Source: www.chegg.com
How can you tell that water has a simple molecular structure? # carbons acyclic alkane # of isomers 4 butane 2 5 pentane 3 6 hexane 5 seven heptane 9 6 contents hide 1 which isomer. Which compound has the highest boiling point?
Source: www.chegg.com
Cycloalkanes have boiling points that are approximately 20 k higher than the corresponding straight chain alkane. Which substance would be expected to have the highest boiling point? The compound with the highest intermolecular forces will have the highest boiling point.
Source: www.chegg.com
80% (217 ratings) sign up for free to view this solution. Remember, the stronger the imf, the higher the boiling point or melting point recall that there are several types of intermolecular forces or imf (ranked from the strongest to the. Alcohol are found to have boiling point approximately as 78.37.
Source: www.chegg.com
Therefore the element with the greatest total number of electrons will have the highest boiling point (iodine) and the element with the smallest total number of electrons will have the lowest boiling point (hydrogen). If you googgled, you may find boiling point of $\ce{hf}$ is $\pu{19.5 ^{\mathrm{o}}c}$, while that of $\ce{hi}$ and $\ce{hbr}$ are $\pu{−35.4 ^{\mathrm{o}}c}$ and $\pu{−66 ^{\mathrm{o}}c}$, respectively..
Source: chemistry.stackexchange.com
Because the halogens are all similar in other ways, you would expect i2 to have the greatest dispersion forces and therefore the highest boiling point (and in fact it does). Typically we can rank intermolecular forces as follows: Which compound has the highest boiling point?
Source: oneclass.com
What compound has the highest boiling point?the hydrogen bond has stronger intermolecular forces. Which metal has the highest boiling point? More energy is needed to separate them than short chain molecules which have only weak forces of attraction for each other.
Source: slideplayer.com
Which compound has the highest boiling point quizlet? Which substance would be expected to have the highest boiling point? Answer choice c is polar but is the only choice not capable of hydrogen bonding and so is eliminated.
Source: www.clutchprep.com
D) ch 3 ch 2 ch 2 ch 2 oh. Due to higher molecular mass. Therefore the element with the greatest total number of electrons will have the highest boiling point (iodine) and the element with the smallest total number of electrons will have the lowest boiling point (hydrogen).
Source: www.numerade.com
Of the diatomic elements (h2, n2, o2, f2, cl2, br2, i2), all have dispersion forces. Carbon has the highest melting point at 3823 k (3550 c) and rhenium has the highest boiling point at 5870 k (5594 c). Which compound has the highest boiling point?
Source: www.chegg.com
The compound with the highest intermolecular forces will have the highest boiling point. Which metal has the highest boiling point? Therefore the element with the greatest total number of electrons will have the highest boiling point (iodine) and the element with the smallest total number of electrons will have the lowest boiling point (hydrogen).
Source: www.bartleby.com
Because the halogens are all similar in other ways, you would expect i2 to have the greatest dispersion forces and therefore the highest boiling point (and in fact it does). If you googgled, you may find boiling point of $\ce{hf}$ is $\pu{19.5 ^{\mathrm{o}}c}$, while that of $\ce{hi}$ and $\ce{hbr}$ are $\pu{−35.4 ^{\mathrm{o}}c}$ and $\pu{−66 ^{\mathrm{o}}c}$, respectively. B) ch 3 ch.
Source: www.bartleby.com
Benzene, an aromatic organic compound which has the boiling point of 80.1 0 c. Therefore the element with the greatest total number of electrons will have the highest boiling point (iodine) and the element with the smallest total number of electrons will have the lowest boiling point (hydrogen). The compound with the highest intermolecular forces will have the highest boiling.
Source: www.chegg.com
The vander waals dispersion forces increase as the length of the hydrocarbon chain increases. Benzene, an aromatic organic compound which has the boiling point of 80.1 0 c. Both hcl and hf have higher boiling points due to hydrogen bonding and should fall between the two extremes.