Which Electron Configuration Is Correct For A Sodium Ion . All of the electrons in the noble gas neon (atomic number 10) are paired, and all of the orbitals in the n = 1 and the n = 2 shells are filled. Within the highest energy level, electrons are removed in order of p then s.
Na+ Electron Configuration (Sodium Ion) - Youtube from www.youtube.com
1s2 2s2 2p5 3s2 what is one example for an electron configuration in the excited state for sodium na. In this video we will write the electron configuration for na+, the sodium ion.
Na+ Electron Configuration (Sodium Ion) - Youtube
In a tof mass spectrometer bromine forms ions with formula [br 2]+ sketch the pattern of peaks you would expect to see in the mass spectrum of a sample of bromine. Therefore the calcium electron configuration will be 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2. In this video we will write the electron configuration for na+, the sodium ion.
Source: www.nagwa.com
The p orbital can hold up to six electrons. Therefore , the electron configuration of the sodium ion is. 1s 2 2s 2 2p 6 3s 2 3p 6 3d 4.
Source: chemistryfromscratch.org
The first is done for you wrong electron configuration correct electron configuration a 1s 2 2s 2 3s 2 3p 2 1s 2 2s 2 2p 4 this is the correct way to do 8 b 1s 2 2s 2 2p 5 3s. The atomic number of sodium is 11. What is the electron configuration notation for sodium?
Source: slidetodoc.com
All of the electrons in the noble gas neon (atomic number 10) are paired, and all of the orbitals in the n = 1 and the n = 2 shells are filled. 1s2 2s2 2p6 3s2 3p2 d. We’ll also look at why sodium forms a 1+ ion and how the electron configurati.
Source: www.youtube.com
In this video we will write the electron configuration for ca2+, the calcium ion. Atoms prefer to gain the stability of octet, by having eight electrons in the outer shell, the electrons of the s and p orbitals. In this video we will write the electron configuration for na+, the sodium ion.
Source: socratic.org
The p orbital can hold up to six electrons. In this video we will write the electron configuration for ca2+, the calcium ion. This list of electron configurations of elements contains all the elements in increasing order of atomic number.
Source: docbrown.info
For atomic br, we have z = 35, and thus for br− we gots 36 electrons to distribute in the usual way. 1s2 2s2 2p6 3s2 3p2 d. 1s 2 2s 2 2p 6 3s 2 3p 6 3d 4.
Source: www.quora.com
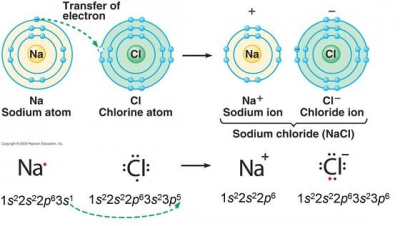
Therefore the calcium electron configuration will be 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2. When sodium (na) and chlorine (cl) are combined, the sodium atoms each lose an electron, forming cations (na+), and the chlorine atoms each gain an electron to form anions (cl−). 1s2 2s2 2p6 3s2 3p1 c.
Source: terpconnect.umd.edu
The total number of electrons is 11 in the sodium atom. Within the highest energy level, electrons are removed in order of p then s. When forming a cation, the electron is removed from the energy level with the highest n value.
Source: slideplayer.com
Calcium electron configuration notation the configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. We’ll also look at why calcium forms a 2+ ion and how the electron configur. We’ll also look at why sodium forms a 1+ ion and how the electron configurati.
Source: slidetodoc.com
These ions are then attracted to each other in a 1:1 ratio to form sodium chloride (nacl). In this video we will write the electron configuration for ca2+, the calcium ion. We'll put six in the 2p orbital and then put the remaining electron in the 3s.
Source: periodictable.me
The m shell contains 3s, 3p, and 3d, and can carry 18 electrons. For na, it is 1s22s22p63s1. 1s2 2s2 2p6 3s2 3p2 d.
Source: www.youtube.com
In this video we will write the electron configuration for ca2+, the calcium ion. Write the electron configuration for sodium. The electron configuration of a neutral sodium atom is #1s^2 2s^2 2p^6 3s^1#.
Source: socratic.org
Thus, its electron configuration is: For na, it is 1s22s22p63s1. The n shell containing 4s, 4d,.
Source: slideplayer.com
What is the electron configuration notation for sodium? One electron is lost to form the sodium ion. We’ll also look at why calcium forms a 2+ ion and how the electron configur.
Source: slidetodoc.com
The electron configurations and orbital diagrams of these four elements are: 1s2 2s2 2p6 3s2 3p2 d. The p orbital can hold up to six electrons.
Source: slideplayer.com
We can represent the electronic configuration as 1s 2 2s 2 2p 6 3s 1. Na + cl ¨ na+ + cl− ¨ nacl. This list of electron configurations of elements contains all the elements in increasing order of atomic number.
Source: www.numerade.com
In the case of sodium the one lone electron in the 3s valence shell would easily be released in order for sodium to have a filled valence shell at 2s22p6. The first is done for you wrong electron configuration correct electron configuration a 1s 2 2s 2 3s 2 3p 2 1s 2 2s 2 2p 4 this is the.
Source: periodictable.me
Write the electron configuration for sodium. Calcium electron configuration notation the configuration notation provides an easy way for scientists to write and communicate how electrons are arranged around the nucleus of an atom. We can represent the electronic configuration as 1s 2 2s 2 2p 6 3s 1.
Source: www.youtube.com
The alkali metal sodium (atomic. These ions are then attracted to each other in a 1:1 ratio to form sodium chloride (nacl). Therefore , the electron configuration of the sodium ion is.
Source: www.slideserve.com
When sodium (na) and chlorine (cl) are combined, the sodium atoms each lose an electron, forming cations (na+), and the chlorine atoms each gain an electron to form anions (cl−). This list of electron configurations of elements contains all the elements in increasing order of atomic number. We’ll also look at why sodium forms a 1+ ion and how the.