Which Element Has The Smallest First Ionization Energy . Which element has the lowest first ionization energy? What kind of element has low electronegativity?
Solved:based On Their Positions In The Periodic Table, Predict Which Has The Smallest First Ionization Energy: Li, Cs, N, F, I. from www.numerade.com
Potassium (option c) has the smallest first ionization energy from the given elements. 1.3k views · answer requested by
Solved:based On Their Positions In The Periodic Table, Predict Which Has The Smallest First Ionization Energy: Li, Cs, N, F, I.
The first chemical element is cesium and the last one is helium. Since the nuclear charge is necessarily diminished with respect to the valence shell, the alkali metals display the lowest ionization energies, and these energies (reasonably) decrease down the group. What kind of element has low electronegativity?
Source: www.quora.com
From the first 18 elements only, choose an element which fits the following descriptions: Since, it has more number of protons. What happens to the values of the successive ionization energies of an element?
Source: www.youtube.com
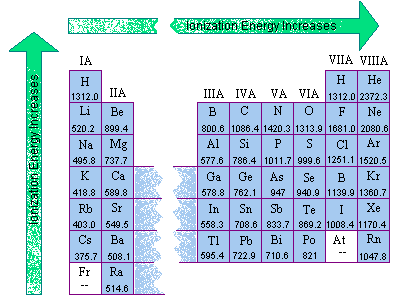
Group 1 of the periodic table features metals whose valence electron singly occupies the outermost shell, the valence shell of the given atoms. As you move across a period, first ionization energy increases. In case of calcium and carbon, calcium will require minimum
Source: physicalsciencetext.weebly.com
What is the first ionization energy? (c) chlorine would have the largest first ionization energy. According to my knowledge, all the alkaline earth elements have the smallest ionization energy.
Source: socratic.org
As you move across a period, first ionization energy increases. I put down cl, but the correct answer is al. Cesium has atomic number 55 and is in the fifth row of the periodic table.
Source: chem.libretexts.org
Ionization energy is the energy required to remove an electron from a gaseous atom or ion. Which element has the smallest ionization energy? The alkali metals have the lowest first ionisation energies in their respective periods of the periodic table because of their low effective nuclear charge and the ability to attain a noble gas configuration by losing just one.
Source: www.numerade.com
Which of the following elements has the smallest first ionization energy? The third ionization energy is the energy it takes to remove an electron from a 2+ ion. Asked aug 28, 2019 in chemistry by carmenon.
Source: examples.yourdictionary.com
104 rows the element which has the highest ionization energy is helium with 24.58741 ev. Group 1 of the periodic table features metals whose valence electron singly occupies the outermost shell, the valence shell of the given atoms. The ionization energy decreases from top to bottom in groups, and increases from left to right across a period.
Source: www.clutchprep.com
The lower the first ionization energy. (2) the 3p electron of aluminium is further from the nucleus compared to the 3s electrons of magnesium. First ionization energy is the energy required to separate one valence electron from an atom in gas phase.
Source: chem.libretexts.org
Cesium has atomic number 55 and is in the fifth row of the periodic table. Thus, helium has the largest first ionization energy, while francium has one of the lowest. (2) the 3p electron of aluminium is further from the nucleus compared to the 3s electrons of magnesium.
Source: www.quora.com
Francium fr (francium) has the lowest ionization energy. Besides, which element has the smallest first ionization energy? All columns align at the bottom.
Source: socratic.org
Cesiumfrom this trend, cesium is said to have the lowest ionization energy and fluorine is said to have the highest ionization energy (with the exception of helium and neon). 104 rows the element which has the highest ionization energy is helium with 24.58741 ev. Now in the case of na and mg ,na has lower ionization energy and mg has.
Source: www.angelo.edu
Besides, which element has the smallest first ionization energy? Group 1 of the periodic table features metals whose valence electron singly occupies the outermost shell, the valence shell of the given atoms. Francium fr (francium) has the lowest ionization energy.
Source: chemed.chem.purdue.edu
As you move across a period, first ionization energy increases. The first chemical element is cesium and the last one is helium. The element with the lowest electronegativity value is francium, which has an electronegativity of 0.7.
Source: www.chegg.com
Which of the following elements has the greatest first ionization energy? Which of these elements has the smallest first ionization energy? The tabular chart on the right is arranged by ionization energy.
Source: www.quora.com
Asked jun 23, 2017 in chemistry by jenny. Since, it has more number of protons. The third ionization energy is the energy it takes to remove an electron from a 2+ ion.
Source: www.clutchprep.com
This value uses the pauling scale to measure electronegativity. If anyone could clarify would be. Which element has the lowest ionization energy?
Source: quizlet.com
A reasonable explanation for this fact involves pairing of two electrons in one 3p orbital in sulfur atoms Besides, which element has the smallest first ionization energy? In case of calcium and carbon, calcium will require minimum
Source: www.angelo.edu
Potassium (option c) has the smallest first ionization energy from the given elements. Which element has the highest first ionization energy? (c) chlorine would have the largest first ionization energy.
Source: www.ck12.org
Cesium has atomic number 55 and is in the fifth row of the periodic table. I put down cl, but the correct answer is al. These include li, na and k.
Source: www.quora.com
The first ionization energy of sulfur is less than that of a phosphorus. The element with the lowest electronegativity value is francium, which has an electronegativity of 0.7. From the first 18 elements only, choose an element which fits the following descriptions: