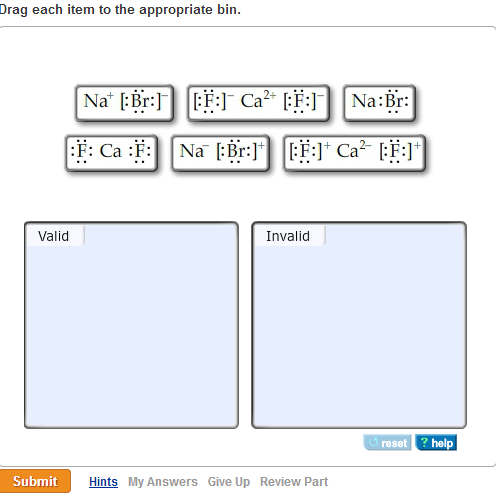
Which Of The Following Are Valid Ionic Lewis Structures . Which nonmetals could form an ionic compound with magnesium with the formula mgx2 (where x represents the nonmetal)? View available hint (e) reset help na ff] br ca br:]| |na:f:
5.1 Lewis Symbols And Structures | General College Chemistry I from courses.lumenlearning.com
Resonance structures for a given compound always contribute equally to the resonance hybrid. Drag each item to the appropriate bin.
5.1 Lewis Symbols And Structures | General College Chemistry I
Which of the following are valid ionic lewis structures? Which of the following is. Which of the following are valid ionic lewis structures?
Source: www.khanacademy.org
Drag each item to the appropriate bin. Which of the following are valid ionic lewis structures? Represent the following ionic compounds by lewis structures:
Source: www.clutchprep.com
Drag each item to the appropriate bin. Which statement below concerning molecular, complete ionic, and net ionic equations, is true? View available hint(s) reset help na [:c1:] [:c1:
Source: www.chegg.com
Valid invalid submit macbook air. These two equivalent structures are referred to as _____ structures. Write a plausible lewis structure for crotonaldehyde, ch3chchcho, a substance used in tear gas and insecticides.
Source: oneclass.com
In both structures, the carbon atoms have a trigonal planar geometry. View available hint (e) reset help na ff] br ca br:]| |na:f: Valid invalid submit macbook air.
Source: itprospt.com
Which of the following are valid ionic lewis structures? Which of the following is the correct lewis structure for the compound pbr 3? View available hint (e) reset help na ff] br ca br:]| |na:f:
Source: www.clutchprep.com
Valid invalid submit macbook air. Which of the following are valid ionic lewis structures? Resonance hybrids contain delocalized electrons.
Source: courses.lumenlearning.com
Which of the following is an ionic compound? Which of the following does not represent a valid reason for globalizing operations? M which of the following are valid ionic lewis structures?
Source: www.coursehero.com
Which of the following is a valid c++ array definition; Represent the following ionic compounds by lewis structures: Which of the following does not represent a valid reason for globalizing operations?
Source: www.youtube.com
Draw the lewis structure for the sulfite ion, so 3 2−. In the lewis structures listed here m and x represent various elements in the third period of the periodic table. View available hint(s) reset help na [:c1:] [:c1:
Source: en.wikipedia.org
Consider the total ionic equation below. Resonance hybrids contain delocalized electrons. View available hint (e) reset help na ff] br ca br:]| |na:f:
Source: www.numerade.com
View available hint (e) reset help na ff] br ca br:]| |na:f: Which of the following is an ionic compound? Valid invalid submit macbook air.
Source: www.chegg.com
Drawing lewis structures where one or more atoms does not have a full valence shell. Drag each item to the appropriate bin. Which of the following is not a valid c++ identifier?
Source: www.chegg.com
Which of the following are valid ionic lewis structures? Lewis structures for ionic compounds ionic compounds contain both a cation and an anion held together by strong electrostatic forces (originating in their charges.) lewis structures for ionic compounds are written by putting two lewis ionic symbols together. Which nonmetals could form an ionic compound with magnesium with the formula mgx2.
Source: www.chegg.com
Drag each item to the appropriate bin. For ionic compounds, the formal charges on the atoms must add up to the charge on the ion. Which of the following are valid ionic lewis structures?
Source: www.chegg.com
S, electrons are transferred esulting in the formation of which of the following are valid ionic lewis structures? Drag each item to the appropriate bin. Represent the following ionic compounds by lewis structures:
Source: shimizu-uofsc.net
The repulsion between these groups produce a linear shape for the molecule with bond angle of 180. Which of the following are valid ionic lewis structures? Drag each item to the appropriate bin.
Source: oneclass.com
In both structures, the carbon atoms have a trigonal planar geometry. Represent the following ionic compounds by lewis structures: Lewis structures for ionic compounds ionic compounds contain both a cation and an anion held together by strong electrostatic forces (originating in their charges.) lewis structures for ionic compounds are written by putting two lewis ionic symbols together.
Source: www.youtube.com
Which of the following are valid ionic lewis structures? Which of the following does not represent a valid reason for globalizing operations? Drag each item to the appropriate bin.
Source: www.kentchemistry.com
Which of the following are valid ionic lewis structures? Which is the net ionic equation for the reaction? See page 637 of your text.
Source: www.clutchprep.com
S, electrons are transferred esulting in the formation of which of the following are valid ionic lewis structures? Drag each item to the appropriate bin. Which of the following are valid ionic lewis structures?